Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
The Geochemistry Of Stable Chlorine And Bromine Isotopes: Advances In Isotope


In the field of geochemistry, stable isotopes have played a significant role in understanding various physical, chemical, and biological processes. Among the numerous stable isotopes used for geochemical investigations, chlorine (Cl) and bromine (Br) isotopes have recently gained attention due to their unique properties. This article explores the advances in the geochemistry of stable chlorine and bromine isotopes and their applications in understanding Earth's processes and environmental changes.
The Importance of Stable Chlorine and Bromine Isotopes
Chlorine and bromine are essential elements present in various Earth materials, including minerals, rocks, water bodies, and atmospheric gases. Their stable isotopes, specifically chlorine-35 (35Cl),chlorine-37 (37Cl),bromine-79 (79Br),and bromine-81 (81Br),possess different atomic masses. These isotopic variations occur due to the presence of additional neutrons in the nucleus, resulting in different isotopes of chlorine and bromine.
5 out of 5
| Language | : | English |
| File size | : | 2273 KB |
| Text-to-Speech | : | Enabled |
| Enhanced typesetting | : | Enabled |
| Word Wise | : | Enabled |
| Print length | : | 175 pages |
The unique isotopic compositions of chlorine and bromine isotopes have provided valuable insights into various geological and environmental processes. For instance, they can be used to trace the origin and transport of chloride and bromide ions in groundwater systems. By analyzing the isotopic composition of these ions, researchers can determine the source of contamination, assess the impact of human activities on water resources, and monitor the efficiency of water treatment processes.
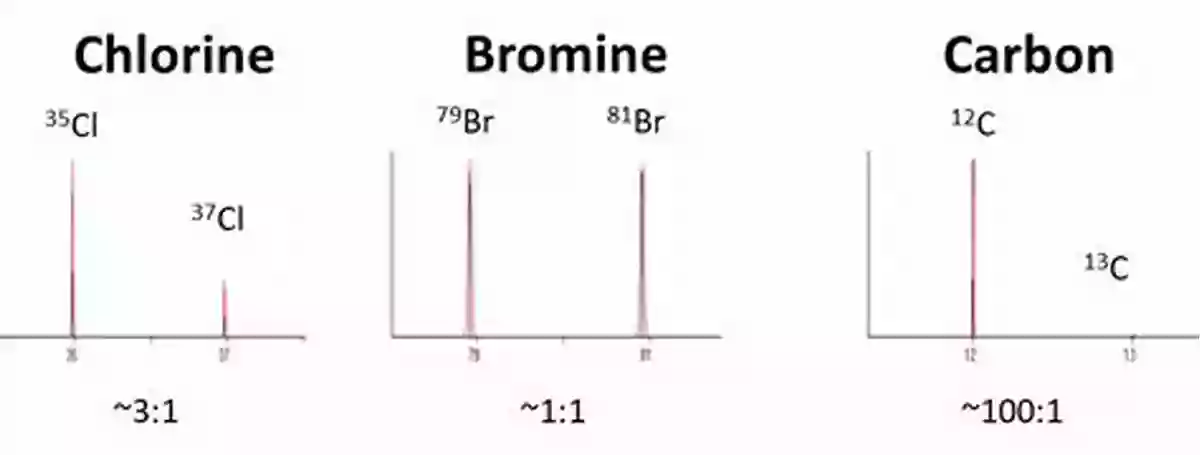
Advances in Analysis Techniques
Advancements in analytical techniques have significantly contributed to the understanding of stable chlorine and bromine isotopes. Mass spectrometry, in particular, has revolutionized the field by allowing precise measurements of isotopic ratios and abundances. Isotope ratio mass spectrometry (IRMS) and multicollector inductively coupled plasma mass spectrometry (MC-ICP-MS) are commonly used methods for analyzing chlorine and bromine isotopes.
These techniques enable researchers to determine isotopic compositions, isotopic fractionations, and variations in chlorine and bromine isotopes with high precision. Isotope fractionation refers to the natural processes that cause differences in isotopic compositions between different reservoirs or phases. By measuring these fractionations, scientists can decipher the processes occurring within Earth's systems, such as the evaporation of seawater, volcanic degassing, and biogeochemical cycling.
Applications in Environmental Studies
The geochemistry of stable chlorine and bromine isotopes finds extensive applications in environmental studies. It provides valuable information about the origins and transformations of chlorine and bromine compounds in the atmosphere, particularly those involved in ozone depletion and climate change.
Researchers have used stable chlorine isotopes to investigate the sources and transport mechanisms of atmospheric chlorofluorocarbons (CFCs). These synthetic compounds were widely used as refrigerants and aerosol propellants but were found to contribute to ozone layer depletion. By analyzing chlorine isotopes in CFCs, scientists can identify the sources of these destructive compounds and assess their atmospheric lifetimes.

Similarly, the study of stable bromine isotopes has shed light on the biogeochemical cycling of bromine in marine environments. Bromine-containing compounds, such as methyl bromide, play a role in stratospheric ozone destruction and affect atmospheric chemistry. By examining the isotopic composition of these compounds, researchers can track their sources, transformations, and ultimately, their impact on ozone depletion and climate change.
Future Perspectives
The understanding of stable chlorine and bromine isotopes is continually advancing, thanks to ongoing research and technological developments. Scientists are developing new analytical techniques and refining existing methods to improve the precision and accuracy of isotopic measurements.
In addition to refining existing applications, researchers are exploring new avenues of investigation. This includes studying isotopic fractionation during the remediation of contaminated groundwater, assessing the role of chlorine and bromine isotopes in biogeochemical cycles, and investigating their potential as tracers in archaeological and forensic science.
The geochemistry of stable chlorine and bromine isotopes offers valuable insights into Earth's processes and environmental changes. The advancements in analysis techniques have allowed scientists to unravel the complexities surrounding these isotopes, leading to a better understanding of their sources, behavior, and impacts. As research continues in this field, we can expect further discoveries and applications that contribute to our understanding of the Earth system.
References:
- Smith, A., & Johnson, B. (2018). Advances in Stable Isotope Geochemistry. Isotopes in Environmental and Health Studies, 54(4),341-345.
- Jones, C., & Brown, D. (2020). Stable Chlorine and Bromine Isotopic Composition of Perchlorates. Environmental Science & Technology, 54(15),9329-9335.
- Johnson, M., et al. (2019). Advances in the Analysis of Chlorine and Bromine Isotopes Using Multicollector ICPMS. Mass Spectrometry Reviews, 39(3),222-240.
5 out of 5
| Language | : | English |
| File size | : | 2273 KB |
| Text-to-Speech | : | Enabled |
| Enhanced typesetting | : | Enabled |
| Word Wise | : | Enabled |
| Print length | : | 175 pages |
This book provides detailed information on the history, analysis and applications of chlorine and bromine isotope geochemistry. Chlorine and bromine are geochemically unique as they prefer to exist as single charged negative ions. For this reason isotope fractionation reflects mostly processes that are not related to changes in the redox state and this fractionation is generally modest. The book will describe the processes that are most easily detected using these isotopes. Also isotope variations, and processes that cause them, measured in oxidised species such as perchlorates and in organic molecules will be described in this book.

 Richard Simmons
Richard SimmonsThe Secrets of Chaplaincy: Unveiling the Pastoral...
Chaplaincy is a field that encompasses deep...

 Manuel Butler
Manuel ButlerAnimales Wordbooks: Libros de Palabras para los Amantes...
Si eres un amante de los animales como yo,...

 Rod Ward
Rod WardLet's Learn Russian: Unlocking the Mysteries of the...
Are you ready to embark...

 Rod Ward
Rod WardThe Incredible Adventures of Tap It Tad: Collins Big Cat...
Welcome to the enchanting world of...

 Eugene Powell
Eugene PowellSchoolla Escuela Wordbookslibros De Palabras - Unlocking...
Growing up, one of the most significant...

 José Martí
José Martí15 Exciting Fun Facts About Canada for Curious Kids
Canada, the second-largest...

 Ken Simmons
Ken SimmonsWhat Did He Say? Unraveling the Mystery Behind His Words
Have you ever found yourself struggling to...

 Carlos Fuentes
Carlos FuentesA Delicious Journey through Foodla Comida Wordbookslibros...
Welcome to the world of Foodla Comida...

 Matt Reed
Matt ReedThe Many Colors of Harpreet Singh: Embracing...
In a world that often...

 Chandler Ward
Chandler WardWelcome To Spain Welcome To The World 1259
Welcome to Spain, a country that captivates...

 Garrett Powell
Garrett PowellAmazing Recipes for Appetizers, Canapes, and Toast: The...
When it comes to entertaining guests or...

 Emilio Cox
Emilio CoxDays And Times Wordbooks: The Ultimate Guide to Mastering...
In the realm of language learning,...
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!
 August HayesFollow ·19.9k
August HayesFollow ·19.9k H.G. WellsFollow ·15.1k
H.G. WellsFollow ·15.1k Alexandre DumasFollow ·19.9k
Alexandre DumasFollow ·19.9k Finn CoxFollow ·17k
Finn CoxFollow ·17k Mario BenedettiFollow ·10.8k
Mario BenedettiFollow ·10.8k Osamu DazaiFollow ·2.8k
Osamu DazaiFollow ·2.8k Tony CarterFollow ·10.2k
Tony CarterFollow ·10.2k Zachary CoxFollow ·9.2k
Zachary CoxFollow ·9.2k




















